 WELCOME TO OPEFE ARCHIVES
WELCOME TO OPEFE ARCHIVES
Subfamily
Serrasalminae or Family Serrasalmidae?
Keeping up with the
scientific changes
By Frank Magallanes, OPEFE
Photo (left) demonstrates
different body forms of the pirambeba S. rhombeus (top)and the piranha
P. nattereri (bottom)
From Frank Magallanes
 The systemic of the Characiformes has long been (and continues
to be) a difficult task to undertake. They were recognized as a homogeneous
group in 1844 by German ichthyologist's Müller and Trochel. The Viennese Kner
and his student Steindachner (between 1858-1915) followed by describing
accurately a number of species without paying much attention to the
classification. It was not until much later that Carl H. Eigenmann (considered
to be the Father of Characoidologist) established the natural classifications of
characins from South America. His principal manuscripts were posted between 1910
and 1927. He was then followed by one of his students Dr. George S. Myers.
Within recent modern times a series of anatomical studies was completed by S.
Weitzman, T. Roberts, William L. Fink, Antonio Machado-Allison and many others
helped establish the critical position of several groups, which led to the
recognition of many families within the suborder (a recognition that would not
have been accepted during Eigenmann's time). The research revealed the
importance, as well as the complexity, of the Characiformes, and is presently
considered as a key-group among teleosts. Included in this huge grouping to be
discussed specifically at this website are the Serrasalmin which includes the
pacus, silver dollars, pirambebas and of course, the true piranhas. The first
authoritative division of the Characoids into several families (16) was done by
S. Weitzman, in Greenwood et al. (1966). The French ichthyologist J. Géry
(1972) would modify this order in a later manuscript.
The systemic of the Characiformes has long been (and continues
to be) a difficult task to undertake. They were recognized as a homogeneous
group in 1844 by German ichthyologist's Müller and Trochel. The Viennese Kner
and his student Steindachner (between 1858-1915) followed by describing
accurately a number of species without paying much attention to the
classification. It was not until much later that Carl H. Eigenmann (considered
to be the Father of Characoidologist) established the natural classifications of
characins from South America. His principal manuscripts were posted between 1910
and 1927. He was then followed by one of his students Dr. George S. Myers.
Within recent modern times a series of anatomical studies was completed by S.
Weitzman, T. Roberts, William L. Fink, Antonio Machado-Allison and many others
helped establish the critical position of several groups, which led to the
recognition of many families within the suborder (a recognition that would not
have been accepted during Eigenmann's time). The research revealed the
importance, as well as the complexity, of the Characiformes, and is presently
considered as a key-group among teleosts. Included in this huge grouping to be
discussed specifically at this website are the Serrasalmin which includes the
pacus, silver dollars, pirambebas and of course, the true piranhas. The first
authoritative division of the Characoids into several families (16) was done by
S. Weitzman, in Greenwood et al. (1966). The French ichthyologist J. Géry
(1972) would modify this order in a later manuscript.
Piranha and Pirambeba
have distinctive body differences which become apparent as maturity sets in.
These differences are the key reason why South American natives call non-true
piranhas by other names. It is only outside South America (or S. A. non-fishing
city dweller's) that the name "piranha" is loosely applied to all
species within the subfamily Serrasalminae.
HISTORICAL IMAGES
COURTESY OF ADRIEN LEROY
GENERAL EXPLANATION
The name Serrasalminae means saw-salmon-family
the saw or serration pertaining to the scutes (or serrated keel) found on
the belly of these fishes. Both carnivorous (single row teeth per jaw) and the
vegetarian (double row teeth per jaw) practice mimicry. Perhaps this has much to
do with the ecological home they inhabit and survival.
The epithet piranha is
perhaps the most over used common name on fishes that could not even be
scientifically called piranhas. It has been used for Serrasalminae vegetarian
fishes and other related forms. In order to properly understand what a piranha
is, one must do some research into common names and how they are applied. The
Piranha Book, edited by Dr. G. S. Myers, (pg 26, TFH Publications
Inc.,1972) covers what the usage of common names should be defined as. I
recommend the research student use that reference to understand common name
usage.
I frequently use the loose
term piranha when I am discussing the "carnivorous" group as
a whole, since this is the name more closely associated with the fish. For
specifics, I prefer the Brazilian pirambeba for the species not genus Pygocentrus.
For the true piranhas placed in genus Pygocentrus I use the epithet
Caribe or Piranha. Caribe pertains to the Spanish Venezuelan piranhas but is a
much more loose application since many of the piranha-like forms are also called
that. But if you get a native fishermen and try to pin him down he will simply
distinguish the more innocuous species with another name or the true caribe as caribe.
The same holds true for epithet piranha. The native fishermen will give
another name for the piranha-like and use the name piranha for the most
dangerous ones in genus Pygocentrus. One last thing, native fishermen
do not use scientific names in describing their fishes, we do! So we
must be careful when attaching a common name to a scientifically described fish.
It has been common practice
for biologists, news media and laymen to describe vegetarian fishes (pacus and
tambaqui) in the genera Colossoma or Piaractus as belonging to
the "piranha family" they are not! Pacus, and piranhas are
all members of the Characidae family that hosts well-over 2,000 species of fish.
The Characidae family (loosely called tetras) are delineated into
groups or subfamilies.
There have been several
unsuccessful attempts to split the sub-family into 2 groups; Mylinae (pacus and
such) and the Serrasalminae (piranhas and associated forms) during the course of
ichthyologic history. Norman (1929) lumped the both groups into one subfamily
naming it Serrasalmoninae, but got the spelling wrong (should have been
Serrasalminae.) His basis was certain characters found on both species. But
the problem was much deeper than simple character assimilation. It was the
advent of Phylogenetic and DNA evidence which now prevents such delineation. DnA
research has also been used to separate the species with results proving
confounding. Some species merged to the surprise of experts. A good case is the
species Catoprion mento (the wimple piranha), this species is
closest (sister) to Pygocentrus in genetic terms. Ichthyologists over the
centuries have kept this particular species separate in its own ranking, but
with genetics it puts it closer to the true piranhas. The wimple piranha
is a good example of a species having the surname "piranha" while it
is NOT a real piranha by definition (see Myers, 1972).
Most recently, genus Metynnis
was discovered to be closely aligned with genus Pygocentrus using
Phylogenetic and DNA sequencing.
Other species like P. denticulata, which is a piranha, have
unique, specialized teeth which help it remove seed husks, much like a pacu's
teeth.
TWO SCIENTIFIC
OPPOSING VIEWS REGARDING CLASSIFICATION
JACQUES GÉRY
Family Serrasalmidae
Classification: Dr. Jacques Géry; The French ichthyologist Jacques Géry
(1972), modified the group into a new family and sub-family adding a sub-genera
which further delineated the piranhas, pirambebas, and the vegetarian silver
dollars and pacus;
|
|
-
genus Serrasalmus
-
sub-genus
Pristobrycon
-
sub-genus
Pygopristis
-
sub-genus
Pygocentrus
-
sub-genus
Serrasalmus
-
sub-genus Taddyella
|
|
-
sub-genus Myloplus
-
sub-genus
Paramyloplus
-
sub-genus
Prosomyleus
-
sub-genus Myleus
-
genus Uiaritichthys
-
genus Metynnis
|
-
genus Acnodon
-
genus Mylesinus
|
Géry split the group
based on several factor's listed below
-
Teeth variable, usually in
more than one row on upper jaw (exceptions occur in Serrasalmidae). Anal fin
moderate or long, with at least 3 unbranched rays and 10 branched ones (with a
few exceptions in certain regressed species).
-
Scales usually cycloid
(with some exceptions) with circuli of caudal (apical) zone parallel or even
divergent with axis of body (except Serrasalmidae).
-
Maxilla reduced, not
toothed; dorsal fin long, with at least 16 rays; usually a predorsal spine and
a series of ventral spines (serrae) (body very compressed, usually disciform;
teeth variable according to diet; scales small, the circuli concentric).
ANTONIO MACHADO-ALLISON
& WILLIAM L. FINK
SCIENTIFIC CLASSIFICATION
PIRANHAS - Use this link.
-
Kingdom: Animalia
-
Phylum: Chordata
-
Subphylum: Vertebrata
-
Superclass: Osteichthyes
-
Class: Actinopterygii
-
Subclass: Neopterygii
-
Infraclass: Teleostei
-
Superorder: Ostariophysi
-
Order: Characiformes
-
Family: Characidae
-
Subfamily: Serrasalminae
Subfamily Serrasalminae
Classification - Dr. William L. Fink & Dr. Antonio Machado-Allison
The most recent evaluation of
the number of species, valid names, alimentary habits and their Phylogenetic
relationship with other groups was investigated by Fink and Machado-Allison
(Fink, 1978; Machado-Allison, 1982a, 1985). Their present findings do not
substantiate the further splitting as revised by Géry (1972). The reasoning
behind this is because Phylogeny is the now the standard norm. The basic issue
is that many of the older classifications, accepted non-monophyletic groups as
valid to be named. There is evidence that some "pacus" are more
closely related to piranhas than other "pacus" ie; that some pacus
share a more recent common ancestor with piranhas than with the other pacus.
That makes the group "pacus" non-monophyletic (actually paraphyletic).
In relation to today's modern Phylogenetic classifications, the older methods of
determining classification (or ranking) is outdated. The older classifications
were not built with the Phylogenetic philosophy in mind. Right now the
evidence on pacu/piranha relationships is equivocal and its doesn't seem to be
useful to revise the previous classifications.
The ranks are entirely
arbitrary--there is no scientific basis for any ranking procedure, so accepting
a group as a subfamily or family is entirely a matter of taste. See below for
further arguments.
CURRENT SERRASALMINAE
SUBFAMILY GENERIC RANKING - 2009
ABSTRACT
Phylogeny of the Serrasalminae (Characiformes) based on mitochondrial DNA
sequences
Previous work (Ortí et al. 1995) based on DNA sequences of mitochondrial (mt)
rRNA genes showed three main groups within the subfamily Serrasalminae:
(1) a basal clade of
herbivores (Colossoma, Mylossoma, Piaractus);
(2) the "Myleus" clade (Myleus, Mylesinus, Tometes);
(3) the "piranha" clade (Serrasalmus, Pygocentrus, Pygopristis,
Pristobrycon, Catoprion, Metynnis). The genus Acnodon was placed as the
sister taxon of clade (1+2). However, poor resolution within each clade was
obtained due to low levels of variation among rRNA sequences.
D-LOOP PHYLOGENY
Complete sequences of the hypervariable mtDNA D-loop are now presented for a
total of 40 taxa representing all genera in the subfamily to address intragroup
relationships. Phylogenetic analyses of these sequences identify the same
groupings as before and provide further evidence to support the following
observations:
(a) the genera Serrasalmus
and Pristobrycon are paraphyletic and form a group that also includes Pygocentrus;
(b) Catoprion, Pygopristis, and Pristobrycon striolatus form a
well supported clade, sister to the group described above in 'a';
(c) distinction of subgenera within Myleus (i.e., Myleus, Prosomyleus,
Myloplus) is not supported;
(d) Mylesinus and Myleus are paraphyletic, since Tometes sp.
is the sister taxon of Mylesinus paraschomburgkii and Mylesinus
paucisquamatus is most closely related to other species of Myleus.
Present taxonomic structure
as accepted by the U.S. systematists et al. (note the different and
much shorter structure from the Géry classification above): The Tree of Life which is
Phylogenetic based is controversial in many ways. It establishes a new order of
animals that would not have been accepted by previous 19th and early 20th
century authors.
The levels at the generic
(binomen) are not subdivided into lower ranking than genus. That is the main
difference with the Géry classification method which does not use Phylogenetic
methods in placing his fishes and the fact he uses subgeneric ranking.

This
cladogram places several vegetarian fishes into the "piranha" clade.
Unthinkable during Eigenmann's time.
NEWEST
INFORMATION REGARDING CREATION OF FAMILY PLACEMENT - 2008
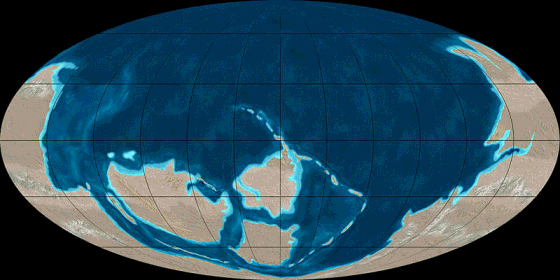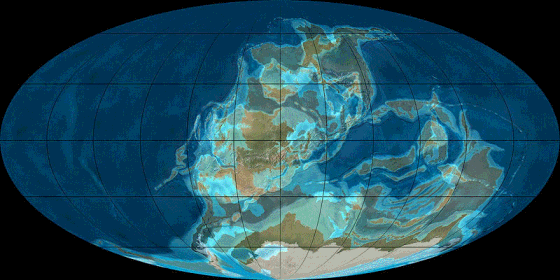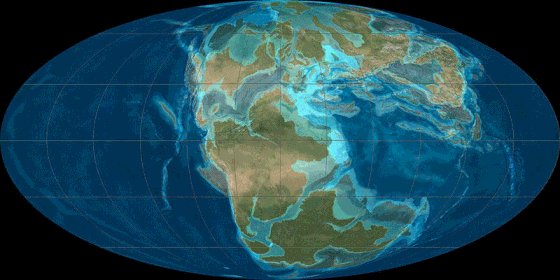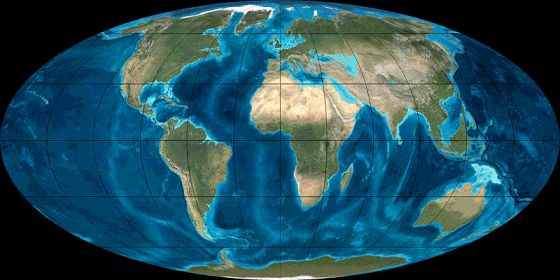
Pangea
Animation
source: http://en.wikipedia.org/wiki/File:TectonicReconstructionGlobal.gif
Phylogeny of the
Serrasalmidae (Characiformes) based on mitochondrial DNA sequences
Guillermo Ortí, Arjun Sivasundar1,
Kelly Dietz and Michel Jégu2
School of
Biological Sciences, University of Nebraska, Lincoln, NE, USA. Genetics and Molecular Biology, 31, 1 (suppl), 343-351 (2008) Copyright
© 2008, Sociedade Brasileira de Genética. Printed in Brazil www.sbg.org.br
ABSTRACT
Previous studies based on DNA
sequences of mitochondrial (mt) rRNA genes showed three main groups within the
subfamily Serrasalminae: (1) a “pacu” clade of herbivores (Colossoma,
Mylossoma, Piaractus); (2) the “Myleus” clade (Myleus,
Mylesinus, Tometes, Ossubtus); and (3) the “piranha” clade (Serrasalmus,
Pygocentrus, Pygopristis, Pristobrycon, Catoprion, Metynnis). The genus Acnodon
was placed as the sister taxon of clade (2+3). However, poor resolution within
each clade was obtained due to low levels of variation among rRNA gene
sequences. Complete sequences of the hypervariable mtDNA control region for a
total of 45 taxa, and additional sequences of 12S and 16S rRNA from a total of
74 taxa representing all genera in the family are now presented to address
intragroup relationships. Control region sequences of several serrasalmid
species exhibit tandem repeats of short motifs (12 to 3 bp) in the 3’ end of
this region, accounting for substantial length variation. Bayesian inference and
maximum parsimony analyses of these sequences identify the same groupings as
before and provide further evidence to support the following observations: (a) Serrasalmus
gouldingi and species of Pristobrycon (non-striolatus) form a
monophyletic group that is the sister group to other species of Serrasalmus
and Pygocentrus; (b) Catoprion, Pygopristis, and Pristobrycon
striolatus form a well supported clade, sister to the group described above;
(c) some taxa assigned to the genus Myloplus (M. asterias, M tiete, M
ternetzi, and M rubripinnis) form a well supported group whereas
other Myloplus species remain with uncertain affinities (d) Mylesinus,
Tometes and Myleus setiger form a monophyletic group.
Key words: piranhas, pacus, D-loop, phylogeny, Bayesian inference. Received:
September 13, 2006; Accepted: April 19, 2007.
Taken from: Phylogeny of the Serrasalmidae
(Characiformes) based on mitochondrial DNA sequences Guillermo Ortí, Arjun
Sivasundar, Kelly Dietz and Michel Jégu, School of Biological Sciences,
University of Nebraska, Lincoln, NE, USA.):
Piranhas and pacus
(Serrasalmids) form a distinctive assemblage of characiform fishes. For a long
time, they were considered a subfamily within the family Characidae. Recent
phylogenetic studies of these fishes, however, strongly suggest that Characidae
is non-monophyletic and that serrasalmids are not closely related to taxa
originally placed in the subfamily Characinae, or other characid subfamilies (Zanata,
2000), but rather that they may be more closely related to Anostomoidea
(Calcagnotto et al., 2005).
All these arguments support
the separate family status of piranhas and pacus; their relationships to other
families within the order Characiformes, however, remain uncertain (Ortí and
Meyer, 1997; Calcagnotto et al., 2005; Hubert et al.,
2005). Species of the Serrasalmidae are endemic to the Neotropics and are
distributed widely in all the major river systems of South America. At least 60
species (in 15 genera) have been recognized. This family includes the well known
piranhas, notorious from accounts of their group predatory behavior, the
seed-eating tambaquí, which is highly regarded as a food species, and the
pacus. Several serrasalmid species are of economic importance and are used in
aquaculture (Junk, 1984; Marshall, 1995; Araujo-Lima and Goulding, 1997) .
Although based on a single molecular marker (mtDNA), the results of this study
carry several taxonomic implications. Most notably, many of the generic
designations in the family seem to lack support or are clearly contradicted by
the data. Some of these conclusions are not new: Pristobrycon striolatus has
previously been regarded as quite distinct from its congeners (Machado-Allison et
al., 1989), differing in several morphological aspects and its
well-supported grouping with Catoprion and Pygopristis is
consistent with the finding of Ortí et al. (1996)(Ortí, Sivasundar,
Dietz and Michel Jégu 2008).
Our present results confirm
this observation and therefore we prefer to restrict Pristobrycon to the
single species P. striolatus, and place all other taxa previously
assigned to this genus in Serrasalmus.
According to the
classification of Géry (1977), the genus Serrasalmus contained the
subgenera Pygopristis, Pristobrycon, Pygocentrus, Taddyella
and the nominate subgenus Serrasalmus; Serrasalmus (Pristobrycon)
striolatus was noted to resemble closely the subgenus Pygopristis.
This observation is well supported by our molecular analysis of control region
data, as this species forms a clade with Catoprion and Pygopristis (Figure
4), and is not closely related to the other specimen putatively assigned to Pristobrycon
(#224 designated Serrasalmus serrulatus here) in the rRNA tree
(Figure 2).
Based on various
morphological characters, Serrasalmus gouldingi is distinct from other
members of the genus (Machado-Allison and Fink, 1996). In this analysis, it was
found to be more closely related to the remaining Pristobrycon than it is
to other species of Serrasalmus. This group containing S. gouldingi,
S. eigenmanni and S. serrulatus is the sister group to the Serrasalmus-
Pygocentrus clade. The genus Serrasalmus contains within it the
genus Pygocentrus.
PRESENT DNA
CONSIDERATION
| |
|
Family Serrasalmidae |
|
|
|
Genus Pygocentrus
Species: piraya,
nattereri and cariba |
Genus Serrasalmus
Species: All other
taxa here |
Genus Pristobrycon
Species: striolatus,
gouldingi(?), eigenmanni(?) and serrulatus(?) |
Genus Catoprion
Species: mento |
Genus Metynnis
Species: Taxa under
consideration |
Results from analysis of
control region sequences of a dense taxonomic sampling for Serrasalmus and
Pygocentrus provides strong evidence for the monophyly of Pygocentrus but
its relationship to diverse components of Serrasalmus remains unresolved
(Hubert et al., 2007). Some of the poor resolution obtained in our study
is evidently the consequence of poor taxonomic sampling (Ortí,
Sivasundar, Dietz and Michel Jégu 2008).
Some authors (e.g. Géry,
1977) have recognized the existence of four subgenera within Myleus,
namely Myloplus, Paramyloplus, Prosomyleus and the nominate
subgenus Myleus, within this genus. These subgeneric distinctions have
been, as with all previous classifications, based primarily on dental
morphology. Other authors, however, rejected these subgeneric distinctions due
to the lack of autapomoprhies (Machado-Allison and Fink, 1995). The monophyly of
subgenera within Myleus is not supported by analyses of mtDNA data.
Analysis of the Myleus group reveals the polyphyly of the formerly
designated genus Myleus and supports the taxonomic rearrangement proposed
by Jégu and Dos Santos (2002) and Jégu et al. (2003), but relationships
among the various components of this group remain tentative. The group formed by
Myleus setiger with Mylesinus and Tometes is
relatively well-supported (PP = 1.00, BV = 67, Figure 3) suggesting strong
affinities of Myleus with species designated to these genera. A robust
group of Myloplus species (M. rubripinnis, M. asterias,
M. tiete, and M. ternetzi) is also well supported by the control
region data. As these analyses have shown, there are several taxonomic
inconsistencies in this subfamily. While this study represents the most
comprehensive molecular systematic treatment of this group, and utilizes a
highly variable mtDNA marker to provide resolution of shallow nodes, placement
of some taxa remains uncertain. In order to provide a strong foundation for
taxonomic revision of the group, future studies would benefit from utilizing
dense taxonomic sampling, nuclear gene sequences, together with mtDNA and
morphological characters (Ortí, Sivasundar, Dietz and Michel Jégu 2008). To
read more about these new revisions contact the authors above or read the .pdf VIEW
Isolation
by distance and Pleistocene expansion of the lowland populations of the white
piranha Serrasalmus rhombeus
NICOLAS
HUBERT, *†§ FABRICE DUPONCHELLE, *‡ JESUS NUÑEZ, *† ROSARIO RIVERA, †
FRANÇOIS BONHOMME § and JEAN-FRANÇOIS RENNO
*†§ *U.R.
175 Institut de Recherche pour le Développement (IRD), GAMET, BP 5095, 361 rue
JF Breton, 34196 Montpellier Cedex 05, France, † Instituto de
Biología Moleculary Biotecnología, Universidad Mayor de San Andres, La Paz,
Bolivia, ‡Unidad de Limnologia y Recursos Aquaticos, Universidad
Mayor de San Simon, Cochabamba, Bolivia, § Laboratoire
Génome, Populations, Interactions, Adaptation,
CNRS-IFREMER-Université Montpellier II, UMR 5171, SMEL, 1 Quai de la Daurade,
34200 Sète, France
ABSTRACT
The genetic variability and
distribution of Amazonian fish species have likely been influenced by major
disturbance events in recent geological times. Alternatively, the great
diversity of aquatic habitat in the Amazon is likely to shape ongoing gene flow
and genetic diversity. In this context, complex patterns of genetic structure
originating from a joint influence of historical and contemporary gene flow are
to be expected. We explored the relative influence of Pleistocene climatic
fluctuations and current water chemistry on the genetic structure of a piranha,
Serrasalmus rhombeus, in the Upper Amazon by the simultaneous analysis of intron
length polymorphism and mitochondrial DNA sequences. The Madeira river is well
suited for that purpose as it is characterized by a great diversity of water
types, the presence of one of the largest floodplain of the Amazon and the
potential occurrence of two Pleistocene refuges. We found evidence of genetic
structure even at a small geographical scale (less than 10 km), indicating that
the floodplain is not a homogenizing factor promoting interdrainage dispersal in
S. rhombeus. Likewise, the hierarchical genetic structure inferred was
correlated to geographical distance instead of habitat characteristic. Our
results also support the hypothesis that the area underwent population expansion
during the last 800 000 years. In addition, a higher level of genetic diversity
was found in the samples from the putative Aripuanã refuge. The present
findings suggest that Pleistocene refuges contributed significantly to the
colonization of the lowlands in the Upper Amazon valley during the Pleistocene.
Keywords: Characidae, museum hypothesis, Neotropics, nonequilibrium,
population expansion, refuge hypothesis
Received 6
November 2006; revision received 20 February 2007; accepted 5 March 2007
To read more about these
revisions contact the authoris above or read the .pdf VIEW
Phylogeography of the piranha genera Serrasalmus and Pygocentrus: implications for the diversification of the
Neotropical ichthyofauna
NICOLAS HUBERT, FABRICE DUPONCHELLE,***
JESUS NUÑEZ, CARMEN GARCIA-DAVILA, DIDIER PAUGY and JEAN-FRANÇOIS
RENNO
Institut de Recherche pour le Développement (IRD), U.R. 175
CAVIAR, 361 rue JF Breton, BP 5095, 34033 Montpellier Cedex 1, France, Département
Milieux et peuplements aquatiques, Unité Biodiversité et dynamique des
communautés aquatiques, US MNHN 0403,
Muséum National d’Histoire Naturelle, 43 Rue Cuvier, 75231 Paris Cedex 05,
France, Institut de Recherche pour le
Développement (IRD), U.R. 131 Biodiversité des grands
Cours d’Eau, 213 rue La Fayette, 75480 Paris Cedex 10, France, Instituto de Investigaciones
de la Amazonía Peruana, Programa de Ecosistemas Acuáticos, Iquitos, Perú,
Instituto de Biología Molecular Biotecnología,
Universidad Mayor de San Andrés, La Paz, Bolivia, **Unidad de Limnología y
Recursos Acuatícos, Universidad Mayor de San Simón, Cochabamba, Bolivia
ABSTRACT
The
phylogenetic relationships within the piranhas were assessed using mitochondrial
sequences with the aim of testing several hypotheses
proposed to explain the origin of Neotropical
diversity (palaeogeography, hydrogeology and museum hypotheses). Sequences
of the ribosomal 16S gene (510 bp) and control region (980
bp) were obtained from 15 localities throughout the
main South American rivers for 21 of the 28 extant piranha species. The results indicate that the genus Serrasalmus
is monophyletic and comprises three major clades.The phylogeographical analyses of these clades allowed
the identification of five vicariant events,
extensive dispersal and four lineage duplications suggesting the occurrence
of sympatric speciation. Biogeographical patterns are
consistent with the prediction made by the museum
hypothesis that lineages from the Precambrian shields are older than those from the lowlands of the Amazon. The vicariant events
inferred here match the distribution of the
palaeoarches and several postdispersal speciation events are identified, thereby matching the predictions of the palaeogeography and
hydrogeology hypotheses, respectively. Molecular
clock calibration of the control region sequences indicates that the main lineages differentiated from their most recent common
ancestor at 9 million years ago in the proto
Amazon-Orinoco and the present rate of diversification is the highest reported
to date for large carnivorous Characiformes. The present
results emphasize that an interaction among
geology, sea-level changes, and hydrography created opportunities for
cladogenesis in the piranhas at different temporal
and geographical scales.
Keywords: Amazon,
biogeography, Brooks parsimony analysis, molecular clock, mtDNA, phylogeny,
piranha
Received 3 August 2006; revision received 20 November 2006; accepted 11
December 2006
To read
more about these revisions contact the authors above or read the .pdf VIEW
J Mol Evol. 2003 Apr;56(4):464-72.
Mitochondrial
genomics of ostariophysan fishes: perspectives on phylogeny and biogeography.
Saitoh
K, Miya M,
Inoue
JG, Ishiguro
NB, Nishida
M.
Source
Tohoku National Fisheries Research Institute, Shinhama, Shiogama 985-0001,
Japan. ksaitoh@affrc.go.jp
ABSTRACT
Ostariophysi is the second
largest superorder within Teleostei. It contains five orders: Gonorynchiformes,
Cypriniformes, Characiformes, Siluriformes, and Gymnotiformes. Resolving the
higher-level relationships among ostariophysan and related fishes will aid in
resolving basal teleostean divergence and provide basis to historical
biogeographic analysis of major freshwater fish groups. In this study, we report
the complete mitochondrial (mt) DNA sequences for eleven ostariophysan fishes
and the results of phylogenetic analyses including these species plus four other
ostariophysan and nine non-ostariophysan teleostean fishes. Maximum likelihood
and maximum parsimony analyses reconfirmed clupeiforms as the closest relatives
of ostariophysans. However, gonorynchiforms were closer to clupeiforms than to
otophysans (ostariophysan groups excluding gonorynchiforms), thus raising a
question over the current definition of Ostariophysi. The lack of clarity in
otocephalan (ostariophysans + clupeiforms) basal relationships implies that such
divergence took place over a short period of time. The monophyly of cypriniforms,
characiphysans (characiforms, siluriforms, and gymnotiforms), and orders or
superorders outside the ostariophysans examined here were conceivably
reconstructed. The phylogenetic hypothesis suggests a Pangean origin of
otophysans. Within characiphysans, gymnotiforms and siluriforms have independent
evolutionary origins and evolutionary histories comparable to or older than that
of characiforms. This helps to explain the present geographic distribution of
characiphysans
Contributors and Advisers
-
Fink, William L.
-
Orti, Guillermo
-
Petry, Paulo
REFERENCES
-
Anderson S, Bankier AT, Barrell BG, de Bruijn
MHL, Coulson AR, Drouin J, Eperon IC, Nierlich DP, Roe BA, Sanger F, et
al. (1981) Sequence and organization of the human mitochondrial genome.
Nature 290:457-465.
-
Aquadro CF and Greenberg BD (1983) Human
mitochondrial DNA variation and evolution: Analysis of nucleotide sequences
from seven individuals. Genetics 103:287-312.
-
Araujo-Lima C and Goulding M (1997) So Fruitful
a Fish: Ecology, Conservation, and Aquaculture of the Amazon’s Tambaqui.
Columbia University Press, New York, 191 pp.
-
Bentzen P, Wright JM, Bryden LT, Sargent M and
Zwanenburg KC (1998) Tandem repeat polymorphism and heteroplasmy in the
mitochondrial control region of redfishes (Sebastes, Scorpaenidae). J Hered
89:1-7.
-
Brown GG, Gadaleta G, Pepe G, Saccone C and
SbisE (1986) Structural conservation and variation in the D-loop-containing
region of vertebrate mitochondrial DNA. J Mol Biol 192:503-511.
-
Calcagnotto D, Schaefer SA and DeSalle R (2005)
Relationships among characiform fishes inferred from analysis of nuclear and
mitochondrial gene sequences. Mol Phylogenet Evol 36:135-153.
-
Cann RL, Brown WM and Wilson AC (1984)
Polymorphic sites and the mechanism of evolution in human mitochondrial DNA.
Genetics 106:479-499.
-
Carpenter J (1988) Choosing among equally
parsimonious cladograms. Cladistics 4:291-296.
-
Eigenmann C (1915) The Serrasalminae and
Mylinae. Ann Carnegie Mus Pittsburgh 9:266-272.
-
Farris JS (1969) A successive approximations
approach to character weighting. Syst Zool 18:374-385.
-
Farris JS, Källersjö M, Kluge AG and Bult C
(1994) Testing significance of incongruence. Cladistics 10:315-319.
-
Farris JS, Källersjö M, Kluge AG and Bult C
(1995) Constructing a significance test for incongruence. Syst Biol
44:570-572.
-
Géry J (1972) Poissons Characoïdes des
Guyanes. I. Généralités. II. Famille des Serrasalmidae. Zool Verhand
122:1-250.
-
Géry J (1977) Characoids of the World. T.F.H.
Publications Inc, Neptune City, 672 pp.
-
Géry J (1984) The fishes of Amazonia. In:
Sioli H (ed) The Amazon, Limnology and Landscape Ecology of a Mighty Tropical
River and its Basin. Junk Publishers, Dordrecht, pp 343-370.
-
GoslineW(1951) Notes on the characoid fishes of
the Subfamily Serrasalminae. Proc Cal Acad Sci ser 4 27:17-64.
-
Goulding M (1980) The Fishes and the Forest:
Explorations in Amazonian Natural History. University of California Press,
Berkeley, 280 pp.
-
Hoelzel AR (1993) Evolution by DNA turnover in
the control region of vertebrate mitochondrial DNA. Curr Opin Genetics
3:891-895.
-
Hubert N, Bonillo C and Paugy D (2005) Does
elision account for molecular saturation: Case study based on mitochondrial
ribosomal DNA among Characiform fishes (Teleostei, Ostariophysii). Mol
Phylogenet Evol 35:300-308.
-
Hubert N, Duponchelle F, Nuñez J, Garcia-Dávila
C, Paugy D and Renno J-F (2007) Phylogeography of the piranha genera Serrasalmus
and Pygocentrus: Implications for the diversification of the
Neotropical ichthyofauna. Mol Ecol 16:2115-2136.
-
Huelsenbeck JP and Ronquist F (2001) MRBAYES:
Bayesian inference of phylogeny. Bioinformatics 17:754-755.
-
Jégu M and Dos Santos GM (1990) Description d’Acnodon
senai n. sp. du Rio Jari (Brésil, Amapá) et redescription d’A.normani
(Teleostei, Serrasalmidae). Cybium 14:187-206.
-
Jégu M and Dos Santos GM (2002) Révision du
statut de Myleus setiger Müller and Troschel, 1844 et de Myleus
knerii (Steindachner, 1881) (Teleostei, Characidae, Serrasalminae) avec
une description complémentaire des deux espèces.
-
Cybium 26:33-57. Jégu M, Keith P and Le Bail
PY (2003) Myloplus planquettei n. sp. (Teleostei, Characidae,
Serrasalminae), une nouvelle espèce de grand Serrasalminae phytophage du
bouclier guyanais (Guyane française). Rev Suisse Zool 110:823-853.
-
Jobb G (2006) TREEFINDER, v. of May 2006.
Munich, Germany, http://www.treefinder.de.
-
Junk WJ (1984) Ecology, fisheries and fish
culture in Amazonia. In: Sioli H (ed) The Amazon, Limnology and Landscape
Ecology of a Mighty Tropical River and its Basin. Dr W Junk Publishers,
Dordrecht, pp 443-476.
-
Kocher TD, Thomas WK, Meyer A, Edwards SV,
Paabo S, Villablanca FX and Wilson AC (1989) Dynamics of mitochondrial DNA
evolution in animals. Proc Natl Acad Sci USA 86:6196-6200.
-
Leite RG and Jégu M (1990) Food habits of two
species of Acnodon (Characiformes, Serrasalmidae) and scale-eating
habits of Acnodon normani. Cybium 14:353-360.
-
Lowe-McConnel RH (1975) Fish Communities in
Tropical Freshwaters: Their Distribution, Ecology and Evolution. Longman,
London, 337 pp.
-
Machado-Allison A (1983) Estudios sobre la
sistemática de la subfamilia Serrasalminae (Teleostei, Characidae). Parte II.
Discusión sobre la condición monofilética de la subfamilia. Acta Biol Venez
11:145-195.
-
Machado-Allison A and Fink WL (1995) Sinopsis
de las Especies de la Subfamilia Serrasalminae Presentes en la Cuenca del
Orinoco. Serie Peces de Venezuela. Museo de Biologia, Caracas, 89 pp.
-
Machado-Allison A and Fink WL (1996) Los Peces
Caribes de Venezuela: Diagnosis, Claves, Aspectos Ecologicos y Evolutivos.
Universidad Central de Venezuela, Caracas, 149 pp.
-
Machado-Allison A, Fink WL and Antonio ME
(1989) Revisión del género Serrasalmus Lacepede, 1803 y géneros
relacionados en Venezuela: I. Notas sobre la morfología y sistemática de Pristobrycon
striolatus (Steindachner, 1908). Acta Biol Venez 12:140-171.
-
Marshall E (1995) Homely fish draws attention
to Amazon deforestation. Science 267:814.
-
Meyer A (1993) Evolution of mitochondrial DNA
of fishes. In: Hochachka PW and Mommsen P (eds) Molecular Biology Frontiers,
Biochemistry and Molecular Biology of Fishes. Elsevier Press, Amsterdam, pp
1-38.
-
Müller K (2005) SeqState - Primer design and
sequence statistics for phylogenetic DNA data sets. Appl Bioinf 4:65-69.
-
Müller K (2006) Incorporating information from
length-mutational events into phylogenetic analysis. Mol Phylogenet Evol
38:667-676.
-
Nelson,
Joseph S. 1994, Fishes of the World, Third Edition xvii + 600, ISSN:
0-471-54713-1, John Wiley and Sons, New York, NY.
-
Nico L and Taphorn DC (1988) Food habits of
piranhas in the low llanos of Venezuela. Biotropica 20:311-321.
-
Norman JR (1929) The South American characid
fishes of the subfamily Serrasalmoninae with a revision of the genus Serrasalmus
Lacepede. Proc Zool Soc London 52:661-1044.
-
Ortí G and Meyer A (1997) The radiation of
characiform fishes and the limits of resolution of mitochondrial ribosomal DNA
sequences. Syst Biol 46:75-100.
-
Ortí G, Petry P, Porto JIR, Jégu M and Meyer
A (1996) Patterns of nucleotide change in mitochondrial ribosomal RNA genes
and the phylogeny of piranhas. J Mol Evol 42:169-182.
-
Palumbi S, Martin A, Romano S, McMillan WO,
Stice L and Grabowski G (1991) The Simple Fool’s Guide to PCR. University of
Hawaii, Honolulu, 46 pp.
-
Posada D and Crandall KA (1998) Modeltest:
Testing the model of DNA substitution. Bioinformatics 14:817-818.
-
Rand DM (1993) Endotherms, ectotherms, and
mitochondrial genome-size variation. J Mol Evol 37:281-295.
-
Ronquist F and Huelsenbeck JP (2003) MRBAYES 3:
Bayesian phylogenetic inference under mixed models. Bioinformatics
19:1572-1574.
-
Saitou N and Nei M (1987) The neighbor-joining
method: A new method for reconstructing phylogenetic trees. Mol Biol Evol
4:406-425.
-
Sambrook J, Fritsch EF and Maniatis T (1989)
Molecular Cloning: A Laboratory Manual. 2nd ed. Cold Spring Harbor Laboratory
Press, Cold Spring Harbor.
-
Starnes, William C , Characiformes -
Research Curator of Fishes, North Carolina State Museum of Natural Sciences,
Research Laboratory, 4301 Reedy Creek Rd., Raleigh, NC, 27607.
-
Swofford DL (2000) PAUP* Phylogenetic Analysis
Using Parsimony (*and Other Methods), v. 4. Sinauer Assoc, Sunderland.
-
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin
F and Higgins DG (1997) The ClustalX windows interface: Flexible strategies
for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876-4882.
-
Winemiller KO (1989) Ontogenetic diet shifts
and resource partitioning among piscivorous fishes in the Venezuelan Llanos.
Env Biol Fishes 26:177-199.
-
Zanata AM (2000) Estudo das relações
Filogenéticas do gênero Brycon Müller and Troschel, 1844
(Characidae, Characiformes).
-
Eschmeyer, W. N. & Fricke, R. (eds.)
Catalog of Fishes electronic version (30 November 2011). VIEW
-
Myers, George S. (1972) - The Piranha Book
(M-539), TFH Publications.
RETURN RESEARCH PAGE
TO RETURN HOME CLICK HERE.
The OPEFE web site and its
contents; is disclaimed for purposes of Zoological Nomenclature in accordance
with the International Code of Zoological Nomenclature, Fourth Edition, Article
8.3 and 8.4. No new names or nomenclature changes are available from statements
at this web site.
Copyright© 1994-2012 Oregon
Piranha Exotic Fish Exhibit (The OPEFE fish exhibit is permanently CLOSED as of
2000) Sutherlin, Oregon. Information posted on this web site is archival data on
fish scientific classifications and other information. DISCLAIMER: The
copyrighted material may not be used for any purpose other than private study,
scholarship or research. Cited information requires credit and this link
www.opefe.com. All rights reserved. All images shown (unless otherwise
noted) is property of OPEFE.

UPDATED: 12/27/2011
 The systemic of the Characiformes has long been (and continues
to be) a difficult task to undertake. They were recognized as a homogeneous
group in 1844 by German ichthyologist's Müller and Trochel. The Viennese Kner
and his student Steindachner (between 1858-1915) followed by describing
accurately a number of species without paying much attention to the
classification. It was not until much later that Carl H. Eigenmann (considered
to be the Father of Characoidologist) established the natural classifications of
characins from South America. His principal manuscripts were posted between 1910
and 1927. He was then followed by one of his students Dr. George S. Myers.
Within recent modern times a series of anatomical studies was completed by S.
Weitzman, T. Roberts, William L. Fink, Antonio Machado-Allison and many others
helped establish the critical position of several groups, which led to the
recognition of many families within the suborder (a recognition that would not
have been accepted during Eigenmann's time). The research revealed the
importance, as well as the complexity, of the Characiformes, and is presently
considered as a key-group among teleosts. Included in this huge grouping to be
discussed specifically at this website are the Serrasalmin which includes the
pacus, silver dollars, pirambebas and of course, the true piranhas. The first
authoritative division of the Characoids into several families (16) was done by
S. Weitzman, in Greenwood et al. (1966). The French ichthyologist J. Géry
(1972) would modify this order in a later manuscript.
The systemic of the Characiformes has long been (and continues
to be) a difficult task to undertake. They were recognized as a homogeneous
group in 1844 by German ichthyologist's Müller and Trochel. The Viennese Kner
and his student Steindachner (between 1858-1915) followed by describing
accurately a number of species without paying much attention to the
classification. It was not until much later that Carl H. Eigenmann (considered
to be the Father of Characoidologist) established the natural classifications of
characins from South America. His principal manuscripts were posted between 1910
and 1927. He was then followed by one of his students Dr. George S. Myers.
Within recent modern times a series of anatomical studies was completed by S.
Weitzman, T. Roberts, William L. Fink, Antonio Machado-Allison and many others
helped establish the critical position of several groups, which led to the
recognition of many families within the suborder (a recognition that would not
have been accepted during Eigenmann's time). The research revealed the
importance, as well as the complexity, of the Characiformes, and is presently
considered as a key-group among teleosts. Included in this huge grouping to be
discussed specifically at this website are the Serrasalmin which includes the
pacus, silver dollars, pirambebas and of course, the true piranhas. The first
authoritative division of the Characoids into several families (16) was done by
S. Weitzman, in Greenwood et al. (1966). The French ichthyologist J. Géry
(1972) would modify this order in a later manuscript.








